"VSports注册入口" Rapid molecular testing or chest X-ray or tuberculin skin testing for household contact assessment of tuberculosis infection: A cluster-randomized trial
- PMID: 40720526
- PMCID: PMC12316388
- DOI: 10.1371/journal.pmed.1004666 (VSports)
Rapid molecular testing or chest X-ray or tuberculin skin testing for household contact assessment of tuberculosis infection: A cluster-randomized trial
Abstract
Background: The World Health Organization recommends evaluation of all household contacts (HHC) of index tuberculosis (TB) patients for TB disease (TBD) and TB infection (TBI). Tests to identify TBI and TBD are preferred but can be skipped in persons living with HIV and children <5 years VSports手机版. There is equipoise on the need for these tests in other HHC. .
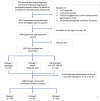
Methods: We conducted a superiority, open label cluster-randomized trial in Benin and Brazil to compare three strategies to evaluate HHC aged 5-50 of persons newly diagnosed with drug susceptible pulmonary TBD: Standard: tuberculin skin testing (TST) for TBI and if positive, chest X-ray (CXR) to rule out TBD; rapid molecular test (RMT): same as Standard, except CXR replaced by an RMT; and No-TST: CXR for all but no TST. Randomization was computer-generated and stratified by country, in blocks of variable length. The primary outcome was TB preventive therapy (TPT) initiation among HHC considered eligible (positive TST, if done, and no evidence of TBD on CXR or RMT). Secondary outcomes were: completion of investigations to detect TBI and TBD, detection of TBD, TPT completion, severe adverse events, and societal costs. V体育安卓版.
Results: Among 1,589 participating HHC enrolled from 29 January 2020, to 30 November 2022, 474 were randomized to the standard, 583 to the RMT, and 532 to the no-TST strategies; all were included in the analyses. Of 848 HHC considered eligible for TPT, 802 (94. 6%) initiated TPT, with no difference between strategies (95%, 94%, and 95% for the standard, RMT, and no-TST strategies, respectively). Of the secondary outcomes, protocol-mandated investigations to detect TBI and exclude possible TBD were completed for 93 V体育ios版. 4% overall, with slight differences between arms (93%, 95%, and 93% for the standard, RMT, and no-TST strategies, respectively). Adverse events resulting in discontinuation of TPT occurred in 3 (0. 4%) participants in total (with 1, 0, and 2 events among participants in the Standard, RMT, and no-TST arms, respectively). The proportion completing TPT was similar with Standard and RMT strategies but was 13% lower (95% confidence interval: 3% to 23% lower) with the No-TST strategy. Societal costs per HHC completing investigations were $61 ($56-$65) with the standard strategy, compared to $52 ($49-$55) with the RMT strategy and $74 ($72-$77) with the no-TST strategy. .
Conclusion: This randomized trial provides high-quality evidence that TST followed by selected use of CXR or an RMT to exclude disease can achieve high rates of TPT initiation at reasonable costs. A limitation of the trial is the potential study effect, which may have affected adherence by providers and HHCs. RMT could replace CXR in the management of HHC in resource limited settings VSports最新版本. .
Registration: clinicaltrials. gov NCT04528823 V体育平台登录. .
Copyright: © 2025 Adjobimey et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited VSports注册入口. .
Conflict of interest statement
The authors have declared that no competing interests exist.
References
-
- World Health Organization. Global Tuberculosis Report 2023. Geneva: World Health Organization. 2024.
-
- Houben RMGJ, Dodd PJ. The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLoS Med. 2016;13(10):e1002152. doi: 10.1371/journal.pmed.1002152 - VSports注册入口 - DOI - PMC - PubMed
-
- United Nations General Assembly. Political declaration of the high-level meeting of the general assembly on the fight against tuberculosis: draft resolution. United Nations Digital Library. 2023;10.
-
- World Health Organization. WHO consolidated guidelines on tuberculosis. Module 1: prevention—tuberculosis preventive treatment. 2nd ed. Geneva, Switzerland: World Health Organization. 2024.
Publication types
MeSH terms
- "V体育平台登录" Actions
- VSports手机版 - Actions
- V体育2025版 - Actions
- "V体育ios版" Actions
- "V体育2025版" Actions
- Actions (VSports最新版本)
- Actions (VSports最新版本)
- "V体育ios版" Actions
- "V体育2025版" Actions
Associated data
- "V体育2025版" Actions
"VSports手机版" LinkOut - more resources
Full Text Sources
Medical