Iron and liver fibrosis: Mechanistic and clinical aspects
- PMID: 30774269
- PMCID: PMC6371002
- DOI: 10.3748/wjg.v25.i5.521
Iron and liver fibrosis: Mechanistic and clinical aspects
Abstract
Liver fibrosis is characterised by excessive deposition of extracellular matrix that interrupts normal liver functionality. It is a pathological stage in several untreated chronic liver diseases such as the iron overload syndrome hereditary haemochromatosis, viral hepatitis, alcoholic liver disease, non-alcoholic fatty liver disease, non-alcoholic steatohepatitis and diabetes. Interestingly, regardless of the aetiology, iron-loading is frequently observed in chronic liver diseases. Excess iron can feed the Fenton reaction to generate unquenchable amounts of free radicals that cause grave cellular and tissue damage and thereby contribute to fibrosis. Moreover, excess iron can induce fibrosis-promoting signals in the parenchymal and non-parenchymal cells, which accelerate disease progression and exacerbate liver pathology VSports手机版. Fibrosis regression is achievable following treatment, but if untreated or unsuccessful, it can progress to the irreversible cirrhotic stage leading to organ failure and hepatocellular carcinoma, where resection or transplantation remain the only curative options. Therefore, understanding the role of iron in liver fibrosis is extremely essential as it can help in formulating iron-related diagnostic, prognostic and treatment strategies. These can be implemented in isolation or in combination with the current approaches to prepone detection, and halt or decelerate fibrosis progression before it reaches the irreparable stage. Thus, this review narrates the role of iron in liver fibrosis. It examines the underlying mechanisms by which excess iron can facilitate fibrotic responses. It describes the role of iron in various clinical pathologies and lastly, highlights the significance and potential of iron-related proteins in the diagnosis and therapeutics of liver fibrosis. .
Keywords: Cirrhosis; Hepatic stellate cells; Iron; Liver fibrosis; Liver pathologies. V体育安卓版.
Conflict of interest statement
Conflict-of-interest statement: All authors declare no conflict of interest.
Figures

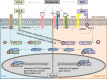
References
-
- Lee UE, Friedman SL. Mechanisms of hepatic fibrogenesis. Best Pract Res Clin Gastroenterol. 2011;25:195–206. - PMC (V体育平台登录) - PubMed
-
- Sharp P, Srai SK. Molecular mechanisms involved in intestinal iron absorption. World J Gastroenterol. 2007;13:4716–4724. - PMC (V体育2025版) - PubMed
-
- Pigeon C, Ilyin G, Courselaud B, Leroyer P, Turlin B, Brissot P, Loréal O. A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload. J Biol Chem. 2001;276:7811–7819. - PubMed
-
- Nemeth E, Tuttle MS, Powelson J, Vaughn MB, Donovan A, Ward DM, Ganz T, Kaplan J. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004;306:2090–2093. - VSports最新版本 - PubMed
Publication types
- Actions (V体育ios版)
MeSH terms
- V体育官网入口 - Actions
- "VSports手机版" Actions
- V体育官网 - Actions
- Actions (VSports app下载)
- VSports最新版本 - Actions
- VSports - Actions
- Actions (VSports在线直播)
- Actions (V体育官网)
Substances
- "VSports注册入口" Actions
- V体育2025版 - Actions
LinkOut - more resources
"VSports注册入口" Full Text Sources
Medical

